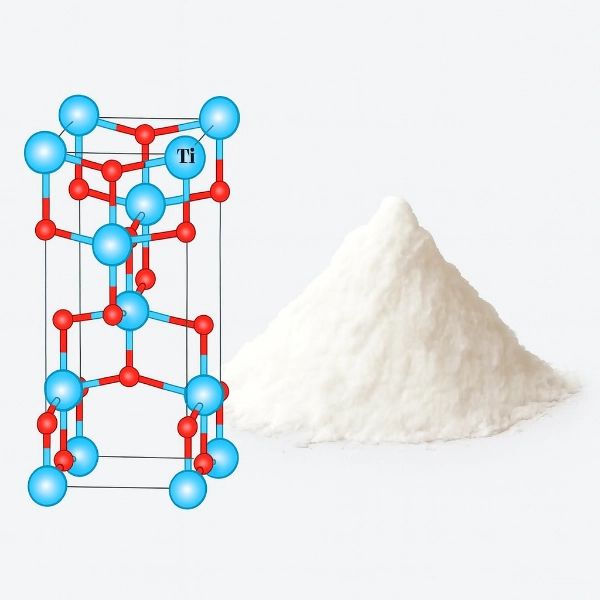
What is the difference between rutile and anatase TiO2?
Rutile and anatase are two different crystal forms of titanium dioxide (TiO₂). They share the same chemical composition, but their crystal structures give them distinct properties and uses.
Rutile is the most stable form, with a higher refractive index and stronger UV resistance. These qualities make it the preferred choice for outdoor coatings, plastics, and other applications that demand long‑lasting whiteness and durability.
Anatase, by contrast, is less stable but has excellent dispersibility and unique photocatalytic activity. Because of these traits, anatase is often used in specialized applications such as self‑cleaning surfaces, solar cells, and certain indoor coatings. It has also been the primary crystal form used in food‑grade titanium dioxide (E171) as a coloring agent, though regulations on food use vary by region.
In brief, rutile is valued for stability and durability, while anatase is chosen for its reactivity, dispersibility, and in some cases food coloring applications.